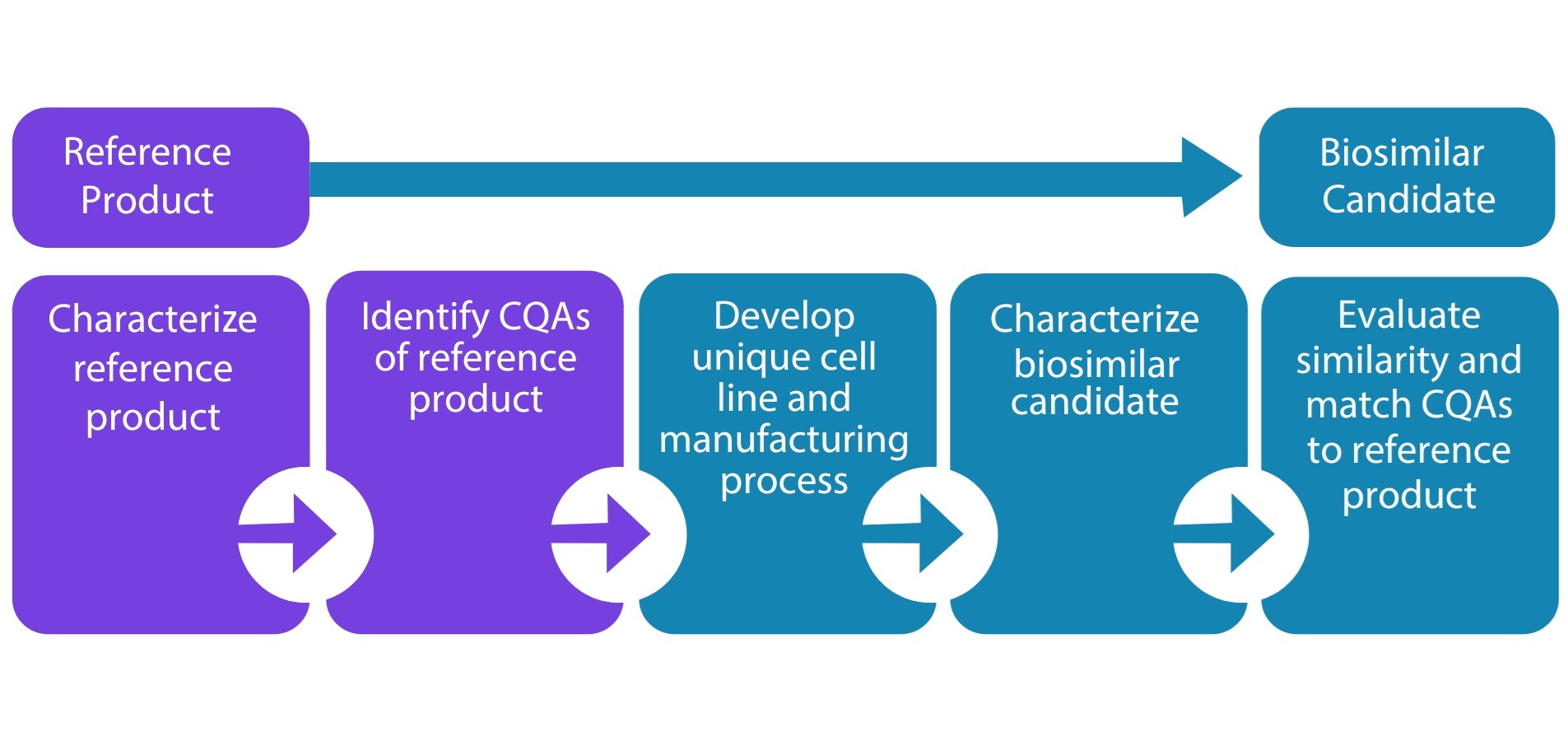
NP-101 Pipeline Drug Development Plan
Novatek pipeline is focused on development of immunomodulatory drugs related to cancers and infectious diseases
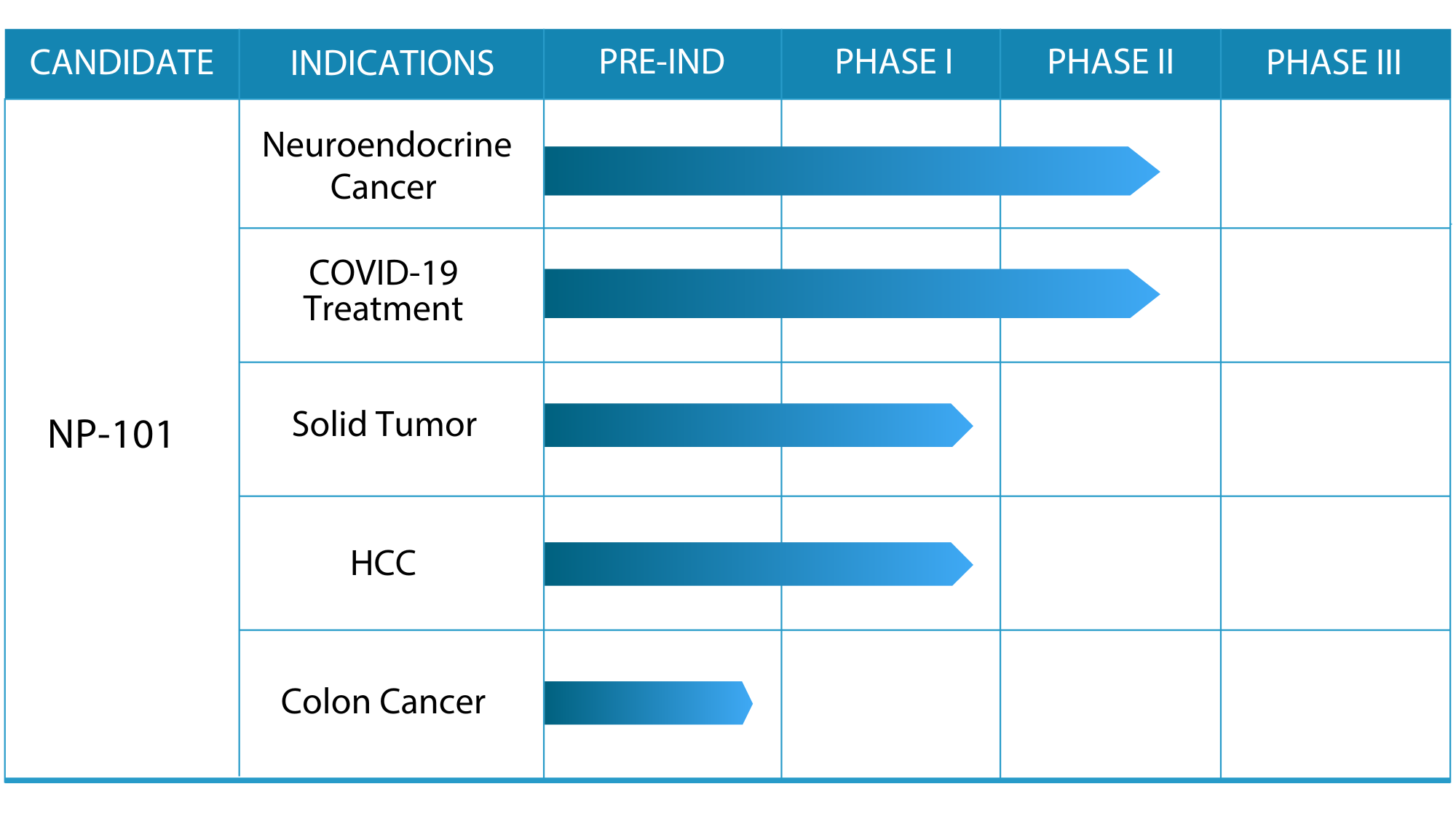
Joint IP Project
BioSimilar Drug:
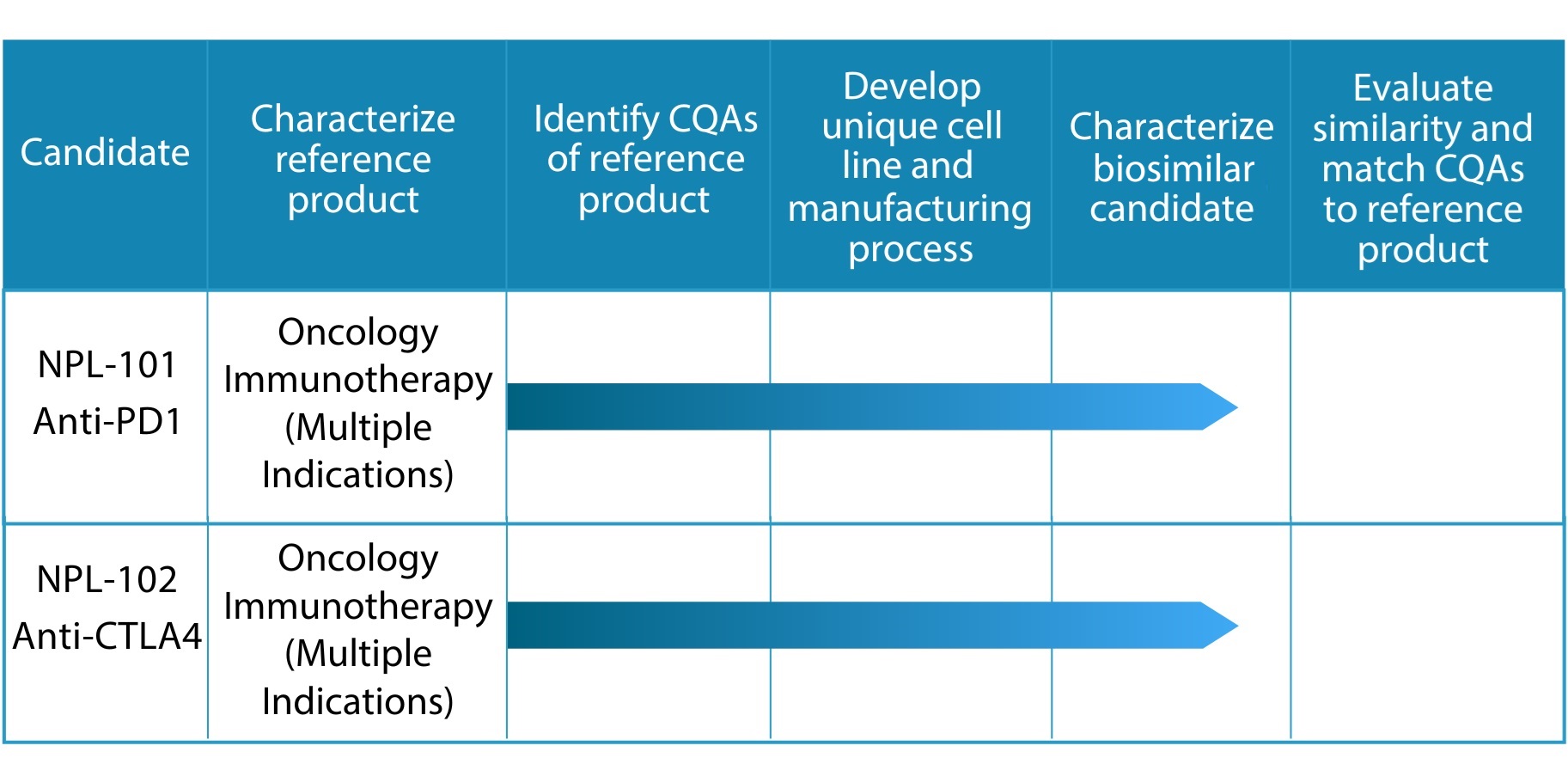
Biosimilars are developed based on information from testing the reference product and matching critical quality attributes (CQAs)